Indy Ryan:
Well, I guess it depends on which reference material you choose to believe.
First, the facts:
That black stain around each nail is something called "Iron Gall Ink", and it's produced whenever the tannins in plants reacts with iron (from the nails). Tannins are common in plants. When you bite down on a grape seed, the astringent taste is due to tannins in the seed. The varying degree of yellow, red and brown colour in red oak, redwood, southern yellow pine, cedar and mahogony is due to tannins in the wood. Tannins are extremely soluble in water, and in fact tea gets much of it's reddish colour from the tannins in tea leaves.
Tannins in turn are "phenols" or "polyphenols". A "phenol" is just a benzene ring with at least one hydroxyl group attached to it, like this:
or
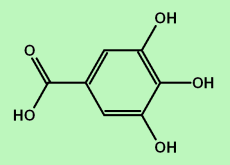
(which is a chemical called "gallic acid")
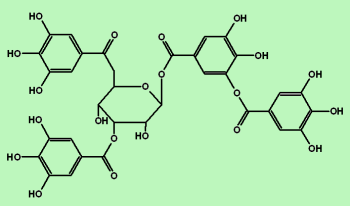
And, if a molecule contains more than one phenol unit, it's called a "polyphenol" like tannic acid:
(Note that tannic acid is really just a central sugar (glucose)molecule with four gallic acid molecules hanging around.)
Well, it turns out that most plants have a lot of polyphenols in them. In fact, it's estimated that about half the mass of a tree's leaf consists of polyphenols.
And, all of those polyphenols are collectively called "tannins". That word "tannin" comes from the fact that these chemicals were used intensively in the leather
tanning industry in the 18 and 1900's.
Tannin - Wikipedia, the free encyclopedia
It turns out that some tannins react with iron or iron ions to form a dark precipitate which is insoluble in water and was used for centuries as writing ink. That precipitate is called "iron gall ink". It was a very popular ink in the middle ages because it is very difficult to remove from paper. In fact, the only way it could be removed from paper was to scrape off the paper containing the ink. At a time when most people couldn't read or write, forgery was a popular way to cheat someone out of house and home by creating false wills, deeds or other documents. Consequently the permanence of iron gall ink made it a favourite choice of notaries, barristers and government officials.
So, on the one hand, most references will tell you to give up on trying to remove iron gall ink from wood or anything else.
However, I am aware of one very reliable reference that says that you can chemically change iron gall ink into a clear salt by reacting it with phosphoric acid or oxalic acid:
Go to the Canadian Wood Council's web site at:
Welcome to CWC
Then hover over the "Publications" link and click on "PDF Publications".
Then, in the "Durability" section, click on the PDF file entitled: "Discolouration on Wood Products: Causes and Implications"
That publication was produced by "Forintek", which is a University of British Columbia research group that investigates problems and recommends solutions for the forest products industry in Canada.
On page 5 of that 8 page pdf file, it talks about non-fungal discolourations on wood, and says:
"Lumber is sometimes discolored with ironstain – this happens when iron particles react with phenolic chemicals in the wood, leaving behind black iron tannates (a common ink pigment). Iron can come from steel wool, filings, lubricants containing metal fines, from invisible iron particles where the wood has rubbed on steel rollers or chains, or even from airborne particles, for example from the brakes or from the wheel-on-rail friction of railway cars (called travel stain). Diagnosing iron stain can usually be done by spotting a dilute (~3%) phosphoric or oxalic acid on the stained part; the acid breaks down the iron tannate into colorless iron salts, and the ironstain is decolorised."
What they're saying is that you can tell if a black discolouration on wood is iron gall ink by treating it with a weak concentration of either phosphoric acid or oxalic acid. If it's iron gall ink, the acid will break the iron tannates down to a colourless salt, thereby removing the stain.
Now, phosphoric acid is commonly found in bathroom and toilet bowl cleaners. That's because it cuts through soap scum like a hot knife through butter, but won't attack chrome. Toilet bowl cleaners will also use phosphoric acid as the active ingredient because it's strong enough to dissolve anything you're likely to find in a toilet bowl while still being relatively mild on both your hands and the environment. The problem with using a toilet bowl cleaner is that it will be gelled so that the acid sticks to the sides of the bowl better and doesn't drain off, and that same gelling will also prevent the acid from being wicked into the wood, which is what you need it to do to get at the iron tannates that are causing the black stain. I'd phone any place listed in your yellow pages under "Janitorial Equipment & Supplies" and ask if they sell a non-gelled phosphoric acid based bathroom cleaner. I know that Buckeye products makes a non-gelled bathroom cleaner called "Sparkle" that's about 40 percent phosphoric acid. Sparkle can be diluted with 12.75 (twelve and three quarters) parts water to make a 3 percent phosphoric acid solution.
Oxalic acid is commonly found in hardware stores. It's sold as the product called "wood bleach" because it's used to remove the yellowing of the wood that occurs when it's exposed to the Sun outdoors.
Maybe ask the home owner if you can try phosphoric acid or oxalic acid on some of those nail holes to see if you can remove the black stains around them. But, maybe don't tell him/her what you used if it works because he/she may want more for the house if they know that the stains on the hardwood floor can be removed.
Hope this helps you.