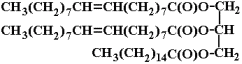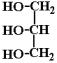at this point I was resigned to try sanding/smoothing the flaked areas again, then sand and TSP the entire interior,
Yeah, I think it needs more work. I plan to sand everything again, then wash with TSP.
You don't need to sand AND wash with TSP. One or the other is fine.
Also, after all the prep, I plan to prime with BM Fresh Start (latex primer, but from what I have learned, it will work great over an oil paint as long as the oil is de-glossed).
Any paint will stick well to an oil based paint if that oil based paint is deglossed.
You should know that TSP was historically used as a cleaner for cleaning walls prior to painting because it deglosses drying oil based paints, which are paints that use a drying oil like boiled linseed oil or Tung oil as the binder. TSP should also degloss real varnish, which is nothing more than plant resins (called "copals") dissolved in drying oils like linseed oil and Tung oil.
You don't need to know the rest, but it explains why I expect a strong concentration of TSP SHOULD degloss alkyd paint (and even alkyd based polyurethane) as well:
All animal fats and vegetable oils (and even animal oils like whale oil) are "tryglycerides", which means that they consist of three "fatty acids" all connected to a glycerine molecule:
In the above depiction of a tryglyceride, there are three fatty acid hydrocarbon chains connected to a glycerine molecule. The glycerine molecule is the one on the right hand side that looks like this:
Now, notice that the two upper fatty acid hydrocarbon chains each have a carbon atom double bonded to another carbon atom? Well, when that happens, then the oil molecule is said to be "unsaturated" because it's possible to add more hydrogen atoms to it. And, those instances of double bonds between carbon atoms are called "unsaturated sites".
Now, when plant oil or animal oil molecules contain a lot of those unsaturated sites, then there's the strong possibility that there will be instances where two unsaturated sites will be in close proximity. When you have two unsaturated sites close by one another, then an O2 molecule from the air will react if it passes close to those unsaturated sites. It will break up into two Oxygen atoms, and each Oxygen atom will react with both unsaturated sites to form a pair of C-O-C crosslinks between the two former unsaturated sites.
Linolenic acid is the fatty acid found most commonly in linseed oil, and it consists of a hydrocarbon chain 18 carbon atoms long with three unsaturated sites in it. So, linseed oil contains lots and lots of unsaturated sites, and when it's exposed to air, it gradually forms those C-O-C crosslinks inside it connecting linseed oil molecules together. The more crosslinking that occurs, the more and more the liquid oil gets thicker and thicker and starts to behave more like a solid than a liquid. Eventually, linseed oil will solidify into a solid as Oxygen molecules from the air spot weld the linseed oil molecules together over time. That process is called "auto-oxidation".
Nowadays, you can no longer buy boiled linseed oil based paints. Nowadays, the only "oil based" paints sold are "alkyd paints". So, what's an "alkyd paint"?
Based on the last few paragraphs, we know that it's those unsaturated sites in plant oil molecules that react with oxygen in the air to gradually transform the liquid oil into a solid, and hence a film of oil on a wall into a solid decorative coating. There are several oils that do that, including linseed oil, Tung (or Chinawood) oil, Safflower oil, Poppy seed oil, Walnut oil and others. Even some oils harvested from fish have enough unsaturated sites to form high quality paint binders.
To make an alkyd resin, what we do is take the fatty acid chains off the triglyceride molecules and through the miracle of modern chemistry, artificially increase the number of unsaturated sites on those fatty acids. But, since we can do that, we don't need to start with an expensive oil like linseed or Tung oil. We can take a cheap plentiful oil like soy bean oil or corn oil, strip off the fatty acid chains, and pump them up on steroids so that they're bristling with unsaturated sites.
Then we take a synthetic resin called "phthalic anhydrite" (which literally means (what you need to add water to to make naphthalic acid, so in simple terms, Kool-Aid crystals might be thought of as "Kool-Aid anhydrite").
And, finally, we add glycerine, which is the small molecule seen drawn above.
And we mix all three in a pot and bring to a boil.
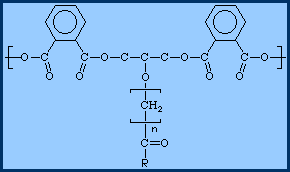
All three of those chemicals will react with the other two, and the result is "clumps" of souped up fatty acid molecules held together with phthalic anhydride and glycerine molecules, and it looks something like this:
where those things that look like crabs are the phthalic anhydride molecules, the short C-C-C chains are the glycerine molecules and those -[CH2]n- things are the souped up fatty acids.
Typically, and alkyd resin will consist of 20 to 30 souped up fatty acids in "clump" held together with the phthalic anhydride and glycerine. We dissolve those alkyd resins in mineral spirits, add coloured pigments for colour and opacity, extender pigments to lower the gloss and increase opacity, and the result is called "alkyd paint".
When you spread alkyd paint on a wall, the first thing that happens is he mineral spirits evaporate. Then, O2 from the air starts to get absorbed into the gazillions of alkyd resins that form a film all over the wall, and the weight of that paint starts increasing because of the absorbed O2 molecules. That auto-oxidation process rapidly increases in speed because of the huge number of unsaturated sites, and so many of them in close proximity. It's that high number of unsaturated sites in the alkyd resins that allow modern alkyd paints to dry much faster and to a much harder film than their old linseed oil counterpart paints did. A modern alkyd paint will be dry to the touch in a few hours as opposed to a few days, and will crosslink much more densely, and that manifests itself in much higher hardness.
But, because both dried linseed oil based paint and dried alkyd paint consists mostly of plant oil fatty acids that have crosslinked together, using TSP to clean dry alkyd paint SHOULD degloss it just like a drying oil based paint. It may take a higher concentration of TSP tho.
Chem 381- CHAPTER THREE- Third Part